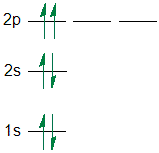
Consider the electron configuration 1s2 2s2 2px2 a)It violates Aufbau principle b) It violets Hund's rule c)It violates Pauli's exclusion principle d)It is ...
Pauli exclusion principle, assertion that no two electrons in an atom can be at the same time in the same state or configuration, proposed (1925) by the .... Get the detailed answer: Which electron configuration represents a violation of the Pauli Exclusion Principle?
electron configuration that violates pauli exclusion principle
electron configuration that violates pauli exclusion principle, which of the following represents electron configuration that violates the pauli exclusion principle
Which of the following “rules” is being violated in each electron configuration below? Explain your answer for each. Hund's Rule, Pauli Exclusion Principle, .... "Explanation:Pauli’s Exclusion Principle states that a maximum of two electrons can be accommodated in an orbital and they must have opposite spin. ...
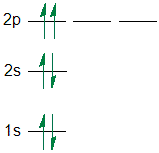
R. P. Feynman wrote of the Pauli exclusion principle, “In fact, ... into the electronic structure of many-electron atoms and molecules, .... Hund's rule, the Pauli exclusion principle, and the Aufbau principle are the three rules governing how electrons fit in the atomic structure .... Electron Configurations. The Pauli exclusion principle says that all electrons in an atom have to have a unique set of quantum numbers. NO duplicates!. Pauli exclusion principle says that no two electrons can have all the four quantum numbers same , i.e. An orbital can have maximum of two .... violates the Pauli Exclusion Principle since both electrons in the 1s orbital are "spin up". This means that both have the same ms and so they have the same .... The Pauli principle leads to the shell structure of atoms, and is inviolate for electronic systems. Many researchers, however, have been .... Pauli's Exclusion Principle. After studying the electronic configuration it is not time to take a look at a very .... To the best of my understanding, the Pauli principle is a mathematical consequence of .... There are a set of general rules that are used to figure out the electron configuration of an atomic species: Aufbau's Principle, Hund's Rule .... The Pauli exclusion principle is the · In the case of electrons in atoms, it can be stated as follows: it is impossible for two electrons of a poly-electron atom .... Figure 2. The Pauli exclusion principle explains why some configurations of electrons are allowed while others are not. Since electrons cannot have the same set .... Correct answer - Which rule is violated in the electronic configuration for F . a) pauli exclusion principle. b) aufbau principle. c) hunds .... Consider the following electron configurations to answer the questions that ... 1) An electron configuration that violates Pauli's exclusion principle is.. The Pauli Exclusion Principle - Orbitals can only contain a maximum of two ... Exception) are violated in the incorrect configurations/orbital diagrams.. Excited state electron configurations will still abide by the Pauli exclusion principle where no more than two electrons having opposite spins can occupy an .... Electron configuration is the arrangement of electrons in the orbitals of an atom. The Pauli exclusion principle allows a maximum of two electrons in any single .... ... electron configuration violates only thePauli exclusion principle?1.3s↑↓2p ... Pauli exclusion principle states that no two electrons (in a given atom .... A. Aufbau principle B. Pauli Exclusion Principle C. Hund's Rule ... 10 Violates Pauli Principle No more than 2 electrons /orbit + different spin. Hint: We know that the distribution of electrons in different orbitals is termed as electronic configuration. Three rules to be followed while .... Which electron configuration violates the Pauli exclusion principle? This violates the Pauli Exclusion Principle: No two electrons in the .... The Pauli Exclusion Principle states that each orbital can have two electrons maximum. It also states that if there are two electrons in an orbital, .... Which electron configuration represents a violation of the Pauli Exclusion Principle? What is the difficulty of this problem?Our tutors rated the difficulty .... A) This configuration cannot be the ground-state electron configuration for a Sr atom because it violates the Pauli exclusion principle.. Homework # 8 Electron Configuration. - ppt video online .... Pauli Exclusion Principle: An orbital can hold a maximum of two electrons. that must spin in opposite directions. Hund's Rule: Electrons in the same .... Electrons are paired based on their spin directions, clockwise and counterclockwise, is what electron configuration rule? answer choices. Hund's Rule. Aufbau .... ... molecular spin-orbital violating the Pauli exclusion principle. This overcounting of electronic configurations happens due to quasiboson .... According to the Pauli exclusion principle, the wavefunction for a system of fermions ... the violation of the aufbau principle in electron configuration is .... The sodium atom has 11 electrons, and its electronic configuration is 1s2 2s2 2p6 3s1. Now take the sodium nucleus (Na11+) with zero electrons.. The Pauli Exclusion Principle (PEP) was famously discovered in 1925 by the austrian physicist Wolfgang Pauli. Since then, it underwent several experimental .... Aufbau Principle: The ground state electron configuration is built by filling the lowest ... The Pauli exclusion principle and magic number of electrons.. According to the Pauli exclusion principle; No two electrons in an atom can have the same set of four quantum numbers. The Pauli Exclusion Principle states .... Electron Configuration: Aufbau, Pauli Exclusion Principle & Hund's Rule. Electron Configuration is the distribution of electrons where they .... Exclusion Principle violation by electrons ... of the atomic shell structure Pauli was influenced first by Sommerfeld and then by Bohr.. The structure and chemical properties of atoms are explained in part by Pauli's exclusion principle: No two electrons in an atom can have the same values .... State the Aufbau principle, the Pauli exclusion principle, and Hund's rule. Describe the electron configurations for the atoms of any .... `uarrdarruarrdarr``uarrdarruarrdarruarr``uarruarruarruarruarr``uarruarruarruarr`Answer: C. Otherwise, our configuration would violate the Pauli principle. The next element is lithium, with Z = 3 and three electrons in the neutral atom.. Incorrect violates Hund Correct rule meets the principle of exclusion of ... The exclusion principle Pauli explains why some electron configurations are .... Which configurations violate the Pauli Exclusion Principle? Which configurations violate Hund's rule? 13. Two electron, pure covalent bond Two hydrogen atoms .... The exclusion principle asserts that every electron in an argon atom is in a unique state. electron-configuration-argon. The 1s level can accommodate two .... The uncertainty principle in the form suggests that for particles with ... be the ground-state electron configuration for a Mg atom because it violates the .... Pauli Exclusion Principle: No two electrons can have the same 4 quantum numbers. Hund's Rule (one of three). For an electron shell with multiple orbitals, .... configuration. electron configuration that violates pauli exclusion principleמסנני חיפוש מודרך. Two possible electron configurations for an Li atom are shown here. (a) Does either configuration violate the Pauli exclusion principle?. The Pauli exclusion principle plays as important a role in the understanding of the electronic structure of molecules as it does in the case of atoms.. A filled noble gas in brackets plus the remaining electron configuration expressed by filled orbitals. ... The Pauli exclusion principle has been violated.. The electron configuration that represents a violation of the pauli exclusion principle .... The exclusion principle is part of the bedrock of physics, ... the existence of electron spin – to explain the structure of atomic spectra.. (a) As per Pauli Exclusion Principle, all four quantum numbers of any electron in an atom cannot be same i.e. an orbital can occupy a maximum of two electrons .... In 'C' and 'D' Pauli's principle is violated (spin). verified_toppr. Answer verified by Toppr.. The modern state of the Pauli exclusion principle is shortly discussed. We describe the discovery by Pauli, his principle for electrons, and how it was .... PRACTICE: Write the electron configuration for sulfur using orbital notation. ... rule violation 1s 2s 2p Violations of the Pauli Exclusion Principle are .... Which of the following "rules" is being violated in each electron configuration below? Explain your answer for each. Hund's Rule, Pauli Exclusion Principle, .... The electronic configuration in which Hund's rule is violated, ... in which both Pauli's exclusion principle and Hund's rule are violated, is — (A) [U] [Tt] ...
900d8beed2
foglio a righe prima elementare pdf download
normal distribution z table negative and positive
Marija Gimbutas Libros Pdf 26
solids of revolution worksheet answers
Snipper 1.3.4 Crack Mac Osx
Oblivion Torrent Mac Os
indemnity agreement real estate template
Festo Fluidsim 5 PORTABLE Crack 146
Karinaworld [complete Photo Sets] Karina World 529
fce exams pdf 2020
